The Ministry of Public Health (MoPH) last night stated that a post-mortem examination on six-year-old Sophia resident, Princess Marcelle Kissoon does not point to any link between the child’s death and the ingestion of the anti-filaria tablet, Albendazole.
Albendazole is one of two tablets currently being distributed by personnel attached to the health ministry, aimed at eliminating lymphatic filariasis.
However, Kissoon’s father of E Field Sophia is claiming that it is this tablet which has caused the death of his only biological child and is imploring the relevant authorities to look into the case.
In light of such, the MoPH when contacted yesterday afternoon, through its Public Relations Officer Terrence Esseboom said, “Our preliminary view does not point to any linkage between the ingestion of the tablet and the child’s death.”

He further noted that Dr. Nancy Olario, the pathologist who conducted the autopsy on the child’s body on Monday, has since asked for time to arrive at the conclusive statement, guided by the samples taken from the deceased’s body.
Roy Kissoon, the bereaved father of the child, told Stabroek News yesterday that he had collected several tablets from the ministry personnel who were distributing the pills, as part of the MDA aimed at eliminating lymphatic filariasis, sometime last week.
Kissoon noted that after sharing with the ministry personnel the names and ages of his household and listening to the instructions on how to administer the medication to his family members, he took the pills home.
He further related, however, that he opted out of administering the pills right away and instead waited until last Wednesday after cooking and eating, to give his daughter along with his foster son, the pills to drink.
“I collected the pills on the road. They were sharing it on the road sometime last week, and we didn’t use it right away. We cook a curry Wednesday, eat the curry and then drink the tablet,” he said.
In relation to questions asked about the dosage given to the now dead child, Kissoon stated that he gave her half of one of the Albendazole tablets, as instructed by the MDA personnel.
The visibly traumatized father went on to say that his daughter seemed fine after taking the pill until later the next day, when he came home and found that she had a “roasting fever,” which later progressed to her having trouble breathing and eating.
“… She didn’t take-in or anything that day, Sister…the next day she rise up and she tell me everything is okay, and Sister, I go out and when I come again she in one roasting fever. She couldn’t talk, and was struggling to breathe. When I reach the hospital I go in and tell them is ah emergency…help me please and they tell me go and sit in the corner and stand up there, and a nurse come and tell me just now. When I carry she in, they told me to wait while they check her, and then they tell me that she gone,” he told Stabroek News.
“But Sister, the only thing that I knowing strange is the tablet; I mine my baby from five months old to six years old, and this is the only thing I know that could have destroyed my baby,” he added.
Asked what he hopes to see done in wake of his daughter’s death, Kissoon said, “I just want some justice for my daughter.”
Additionally, the family is asking for assistance to facilitate the child’s burial since the father is employed on a seasonal basis, and the family is now trying to raise the necessary funds to bury the little girl.
Safe
In its statement, the Ministry said it is confident that the drugs being administered in the Filaria campaign are safe.
It said that the Global Programme to Eliminate Lymphatic Filariasis began in 2001 in Guyana.
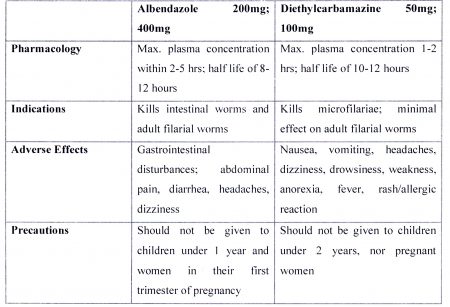
“Since the year 2008, 6 regions have benefitted from the Mass Distribution of the tablets Diethylcarbamazine (DEC) 100mg and Albendazole 400mg at predefined doses. In 2016, 277,612 persons from regions 3, 4, 5 and 10 received the drugs via directly observed therapy (DOT).
Prior to this initiative, thousands of Guyanese received Diethylcarbamazine in DEC salt. The medicines being distributed are donations from the World Health Organization through their regional office: -the Pan American Health Organization”.
The donation process begins with the exchange of documents which include:
a certificate of good manufacturing practice (issued by the Ministry of Health in the country where the tablets are manufactured)
a certificate of analysis of the tablet which assures product quality assurance
an information leaflet on the tablet which details its indications, considerations and precautions, dosage and side effects
The aforementioned documents are first perused by the Permanent Secretary and Chief Medical Officer of the Ministry of Public Health before the drugs are approved for shipment, the statement said.





